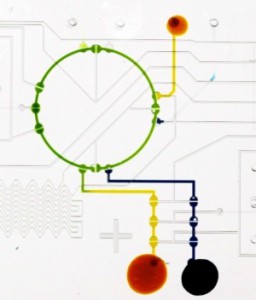
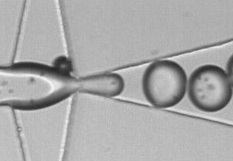
CADMIM goals will be achieved via three coordinated basic and applied research thrust areas:
| (1) “Manufacturable” processes and materials: Many materials currently being used for microfluidic devices are not suitable for large scale production. Fortunately, several manufacturable processes have yet to be explored for the production of low-cost microfluidic chips. Automated roll-to-roll methods for tape-based plastic hot embossing, paper printing, and thin-film metal lamination (i.e. flexible circuit technology), commonly used in consumer products, can all be adapted for “lab-on-a-chip” production. Also, these processes can be merged to create a new class of mass-produced diagnostic devices.(2) Sample Processing and Detection: While these have been issues in the microfluidics field for many years, no group has attempted to jointly address them in the context of mass-produced, low-cost/disposable, easy-to-use diagnostics. Innovation in sample filtering, constituent enrichment and separation, on-chip reagent storage, and fluidic functions (metering, mixing, transport, etc.) in low-cost manufacturable biochips are some of the critical aspects for self-contained labs-on-a-chip. Innovation must also occur at the assay and detection level, such as the development of simplified cellular and molecular assays, tests that implement different transduction mechanisms (e.g. optical, mechanical, magnetic), and probes specifically designed to enable cheap disposable microfluidic platforms with more complex functions.
(3) Integration and control systems: Integration encompasses output from Thrust 1 and 2, namely the combined basic science and implementation of sensing, analysis, and detection in manufacturable biochips. This thrust also includes the development of interfaces to the real world — once results are available they need to be managed and communicated. Options include a visual readout (such as a color change on the chip that can be read by the naked eye or digitally photographed and uploaded to the internet), or an electrical signal that can be interfaced to a smart phone or transmitted via a USB interface to a laptop computer. This thrust also embraces the task of on-command, programmable multiplexed diagnostics for which intelligence is incorporated onto the biochips. |
The three thrust areas are intertwined, and the ultimate challenge is to innovate in all these areas simultaneously to refine on-chip functions, ensure compatibility and connectivity, integrate intelligence and communication, anticipate, overcome or circumvent bottlenecks, and create high capability, self-contained, manufacturable microdevices. Furthermore, new design methodologies and tools will need to be developed no only to support this type of innovation, but also to allow users outside the center to easily design and fabricate scalable diagnostic labs-on-a-chip.
This is a fresh approach to microfluidic biochip development, targeting in advance a dramatic reduction in cost, with equal or superior performance to lab-based functionality, allowing low-cost manufacture and widespread deployment of biomedical microdevices.
Currently funded research projects:
Y10-01: “Hydrostatic Poration of Cells for Molecular Delivery”
David Eddington, Zhangli Peng (U. Illinois Chicago)
Y10-04: “Integrated Multi-Tissue 3D Cell Culture Platform”
Ian Papautsky, Salman Khetani (U. Illinois Chicago)
Y11-01: “Protoplast-based assay for infection-competent viruses within seed samples”
Jered Haun (UC Irvine)
Y11-02: “Arrayed-Droplet based Optical Projection Tomography for CHO Cell Antibody screening (ADOPT-CHO)”
Abraham Lee (UC Irvine)
Y11-03: “Advanced fluorescence microscopy pipeline for a less-invasive sperm quality screening”
Michelle Digman, Francesco Palomba (UC Irvine)
Y11-04: “Focusing of Sperm in Inertial and Viscoelastic Flows”
Ian Papautsky, Zhangli Peng (U. Illinois Chicago)
Y11-05: “Arrayed-Droplet based Optical Projection Tomography for Protoplast Health Screening (ADOPT-PRO)”
Jered Haun, Abraham Lee (UC Irvine)
Concluded research projects:
Y10-02: “Microfluidic Protein Aggregation Gradient Generator (µPrAGG)”
Abraham Lee, Michelle Digman (UC Irvine)
Y10-03: “Microwells with Viability Assessment for 3D Cell Culture”
Ian Papautsky (U. Illinois Chicago)
Y9-002: “Droplet-based Immune Cell Engineering (DICE)”
Abraham Lee (UC Irvine)
Y9-008: “Microfluidic Platform for High-Throughput Transfection of Protoplasts and Other Primary Cells”
Jered Haun (UC Irvine)
Y9-009: “Numerical and Experimental Investigations of Asymmetric Cell Positioning in Inertial Flows”
Ian Papautsky, Zhangli Peng (U. Illinois Chicago)
N-009: “Protoplast-based assay for infectious plant viruses within seed samples”
Jered Haun, Elliot Hui (UC Irvine)
N-008: “Evaluating Microwell Plates for 3D Cell Culture”
Ian Papautsky (U. Illinois Chicago)
Y8-004: “Drug Screening via Miniaturized Cancer Organoids”
Elliot Hui, Chris Halbrook (UC Irvine), David Eddington (U. Illinois Chicago)
Y8-006: “Microfluidic Viscometer by Acoustic Streaming Transducers (µVAST)”
Abraham Lee, Michelle Digman (UC Irvine)
Y8-007: “Electrochemical Interdigitated Array Sensors”
Ian Papautsky (U. Illinois Chicago)
Y8-002: “Large Scale Dynamic Droplet Cell-Cell Pairing Device (D2C2)”
Abraham Lee, Michelle Digman (UC Irvine)
Y7-003: “Precise Cellular Alignment and Positioning in Viscoelastic-Inertial Flows”
Ian Papautsky, Zhangli Peng (U. Illinois Chicago)
Y7-005: “Low-Shear Organoid Vortex Array (LOVA)”
Elliot Hui, Medha Pathak (UC Irvine), David Eddington (U. Illinois Chicago)
Y7-006: “Microfluidic Isolation of Protoplasts for Plant Diagnostics & Gene Editing”
Jered Haun (UC Irvine)
Y6-003: “A Scalable 3D Human Liver Platform for Drug Discovery”
Salman Khetani, David Eddington (U. Illinois Chicago)
Y6-004: “Compound Screening of Cell-Cell Pairing”
Abraham Lee, Michelle Digman (UC Irvine)
Y6-006: “A Microfluidic 3-Part Differential Sorter”
Abraham Lee (UC Irvine)
N-006: “Microfluidic Chips for Cell Positioning and Alignment”
Ian Papautsky (U. Illinois Chicago)
N-007: “NanoIDAs for Electrochemical Sensing”
Ian Papautsky (U. Illinois Chicago)
Y6-005: “Device Design and Protocol Optimization for Dissociation of Diverse Tissues and Organs into Single Cells”
Jered Haun (UC Irvine)
Y4-006: “Droplet KASP Array”
Elliot Hui (UC Irvine)
Y5-005: “Electrochemical Sensors for Plant Nutrition”
Ian Papautsky (U. Illinois Chicago)
Y5-004: “Multi-omic Molecular Identification of Single Cells in Addressable Microwell Arrays”
Jered Haun, Abraham Lee, Wendy Liu (UC Irvine)
Y5-007: “High-content Screening Platform for Multicellular Systems”
Salman Khetani (U. Illinois Chicago), Elliot Hui (UC Irvine)
Y5-008: “Sorting Blood Components”
Abraham Lee (UC Irvine)
Y5-009: “Microfluidic Chips for Electrochemical Metal Extraction”
Ian Papautsky, Jie Xu (U. Illinois Chicago)
N-004: “Electrochemical methods for improving metal extraction”
Ian Papautsky (U. Illinois Chicago)
N-005: “Integrated Tumor Tissue Processing Chip to Generate Single Cells”
Jered Haun (UC Irvine)
Y4-001: “High Density Microfluidic Logic”
Elliot Hui (UC Irvine)
N-003: “Optical Sensing for Stress Biomarker Detection in Bodily Fluids”
Andrew Steckl (U. Cincinnati)
Y4-005: “The Ultimate Sample Preparation for Whole Blood”
Abraham Lee (UC Irvine)
Y4-009: “Nanofluidic Pre-concentration”
Jason Heikenfeld (U. Cincinnati), Kevin Plaxco (UC Santa Barbara)
Y3-002: “Droplet-based Single Cell Assays – from Phenotype to Genotype”
Abraham Lee (UC Irvine), Michelle Digman (UC Irvine), Chong Ahn (U. Cincinnati)
Y3-003: “Rapid Nucleic Acid Purification and Sequence-Specific Detection for Agricultural Applications”
Michelle Khine (UC Irvine), Juan Santiago (Stanford University)
Y3-006: “A Simple Laminated Membrane Approach for Interdigitated Electrodes: Both Easier Integration and Improved Performance for Microfluidic Systems”
Jason Heikenfeld (U. Cincinnati)
Y2-002: “NFC-Operated Quantitative Point-of-care Diagnostic Assays with Low-to-Zero On-Board Power”
Andrew Steckl (U. Cincinnati)
Y2-003: “Tissue Processing Chip for Single-Cell Molecular Diagnostics”
Elliot Hui, Jered Haun (UC Irvine)
Y2-004: “World’s 1st Sweat Stimulation and Sensing Patch! + No Interference! + 170% FOM!”
Jason Heikenfeld, Gerald Kasting (U. Cincinnati), Michelle Khine (UC Irvine)
ES-001: “A Parallel, High Speed Microfluidic Droplet Platform for Multiplexed, High Throughput, and Scaled-Up Biomolecular Assays”
Abraham Lee (UC Irvine), Chong Ahn (U. Cincinnati)
ES-002 “The Microfluidics of On-Skin Technologies”
Jason Heikenfeld, Gerald Kasting (U. Cincinnati)
ES-004: “Nucleic Acid Purification and Sequence-Specific Detection on a Handheld Printed Circuit Board Platform”
Mark Bachman (UC Irvine), Juan Santiago (Stanford University)
ES-006: “Integrated Microfluidics for Isolation and Enrichment of Rare Cells”
Abraham Lee (UC Irvine), Ian Papautsky (U. Cincinnati)
ES-008 “Microfluidics Integration with Mass-Manufactured Paradigms for Flexible and/or Wearable Electronics”
Jason Heikenfeld, Andrew Steckl (U. Cincinnati)
ES-010 “Automated DNA Library Preparation Chip for Next-Generation Sequencing”
Elliot Hui (UC Irvine)
N-001 “Integration of Blue-tooth and Power with Flexible, Conformal Motion Sensors”
Michelle Khine, Mark Bachman (UC Irvine), Jason Heikenfeld (U. Cincinnati)
N-002 “Integration of Electrochemical Impedance Spectroscopy and Multiplex Microfluidics”
Ian Papautsky (U. Cincinnati)